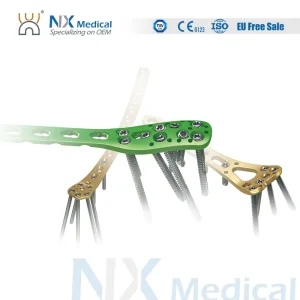
Guangzhou Clerivida Medical Co., Ltd. stands as a premier beacon of innovation in the global orthopedic sector. Specializing in high-precision Elastic Intramedullary Nails (EIN) and comprehensive surgical systems, we integrate advanced R&D with a robust manufacturing ecosystem. Our 18-year legacy in the medical device industry has allowed us to master the delicate balance between biological fixation and biomechanical stability.
As a leading Elastic Intramedullary Nail manufacturer, we address the critical needs of pediatric trauma and long bone fractures. Our systems are designed to provide dynamic stability while promoting rapid callus formation, a fundamental requirement in modern "minimally invasive" orthopedic surgery. We operate with a perfect quality management system, ensuring that every titanium and stainless steel implant surpasses international safety benchmarks.

Unlike rigid nailing, elastic intramedullary nails (TEN/EIN) utilize the principle of three-point fixation. This allows for controlled micro-motion at the fracture site, which significantly accelerates secondary bone healing through external callus formation. It is the gold standard for pediatric femoral and humeral fractures.
We utilize Medical Grade 5 Titanium (Ti-6Al-4V ELI) and 316L Stainless Steel. These materials offer superior biocompatibility and the necessary modulus of elasticity to prevent "stress shielding," a common complication with overly stiff implants.
Our instrumentation is designed for percutaneous insertion, minimizing soft tissue damage and blood loss. This leads to shorter hospital stays and faster rehabilitation cycles for patients, particularly active children and adolescents.

The China medical device supply chain has evolved into a global powerhouse of efficiency and precision. By centralizing our production in Guangzhou, Clerivida leverages:
Navigating the complex regulatory landscape of the medical industry requires more than just a certificate; it requires a culture of quality.
Our factories adhere strictly to ISO 13485:2016 quality management systems. We provide full traceability for every batch of elastic nails, ensuring that hospital procurement teams meet their local health authority requirements (CE, FDA-ready documentation, etc.).
We don't just ship products; we provide surgical protocol training and localized marketing support. Our professional after-sales team works across time zones to ensure that surgeons are confident in the application of our systems.
Understanding that anatomical requirements differ across populations, we offer customization in nail diameters (1.5mm to 4.5mm) and lengths to suit specific regional clinical needs.
The orthopedic industry is shifting towards Digital Surgery and Patient-Specific Implants (PSI). Clerivida is at the forefront, exploring 3D-printed surgical guides and bio-resorbable elastic materials that eliminate the need for secondary implant removal surgeries. Our R&D focuses on "Smart Implants" that can provide feedback on bone healing progress, further enhancing the surgeon's ability to provide data-driven patient care.