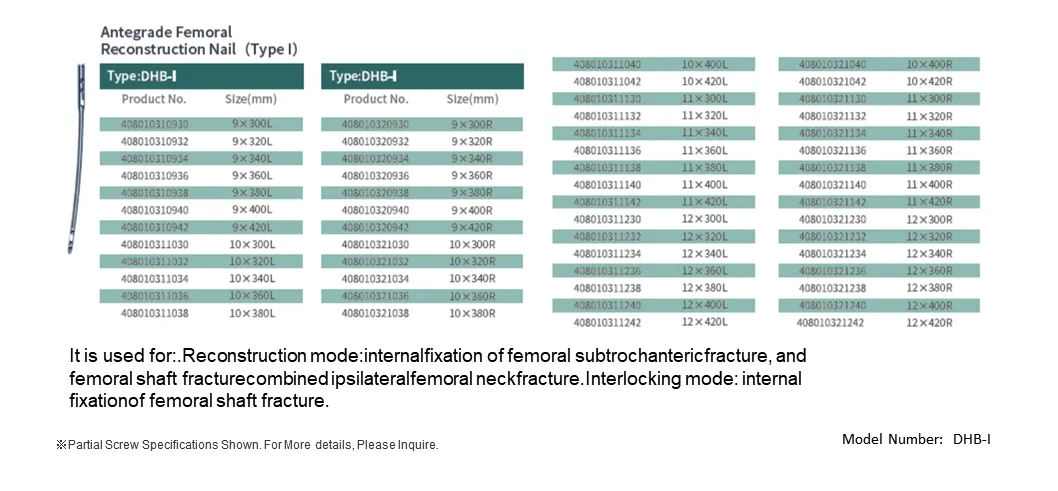
The intramedullary nail system is designed to be implanted within the medullary canal, forming a composite structure with the bone to share mechanical loads. Positioned along the central axis of the femoral shaft, the nail offers optimal resistance to bending forces. Proximal and distal locking screws ensure stable fixation, allowing axial compression while preventing rotational instability. This femoral reconstruction nail system, composed of the main nail, traction screw, locking screws, and end cap, is made entirely from TC4 titanium alloy. It is primarily intended for femoral shaft fractures and complex femoral injury patterns.