





















Established in 1958, the enterprise is specialized in the development, manufacture, and sales of orthopedic implants and instruments. As a key enterprise for national medical apparatus and a director member of the China Medical Apparatus Industry Association, the focus remains on high-quality orthopedic solutions. The product line covers Spine pedical screws, Interlocking nails, Locking plates/screws, trauma plates/screws, cannulated screws, external fixators, hip prosthesis, and surgical instruments.
Products are characterized by multi-varieties and complete specifications. All medical apparatus have passed rigorous approval and authentication from national and provincial superintendent offices. Each product is marked with specialized material codes and laser anti-fake marks, backed by quality responsibility insurance.
Equipped with advanced imported CNC machines and high-precision digital control systems, the facility includes a 100,000 Grade axenic purification chamber to ensure product integrity. The company was among the first to achieve ISO 9001 quality system certification. Our medical apparatuses have been recognized with numerous provincial and national innovation awards for science and technology excellence.
Adhering to the tenet of "creating high quality with science and technology," we pursue perfect design, innovative technology, and complete professional service. We maintain a strict focus on manufacturing standards to serve the global medical community.

We are a dedicated manufacturer of orthopedic implants and instruments since 1958. Our primary expertise includes trauma plates, interlocking nails, spinal fixators, and external fixators.
All products are produced according to EU CE standards. We implement strict protocols, including pre-production samples before mass production and a thorough final inspection before every shipment.
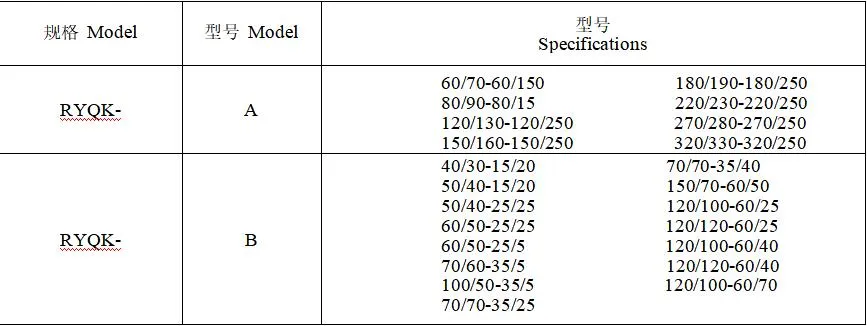
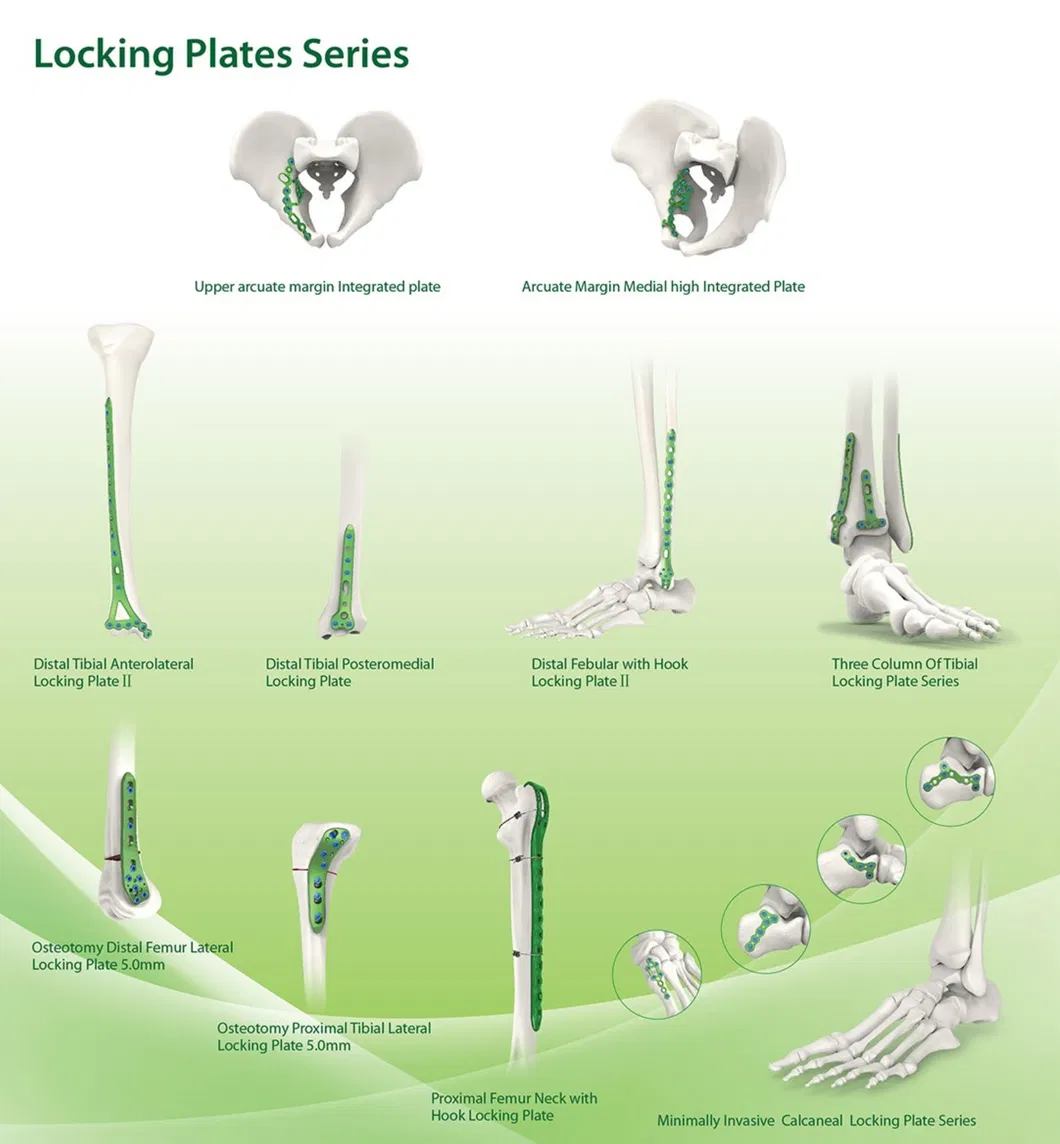
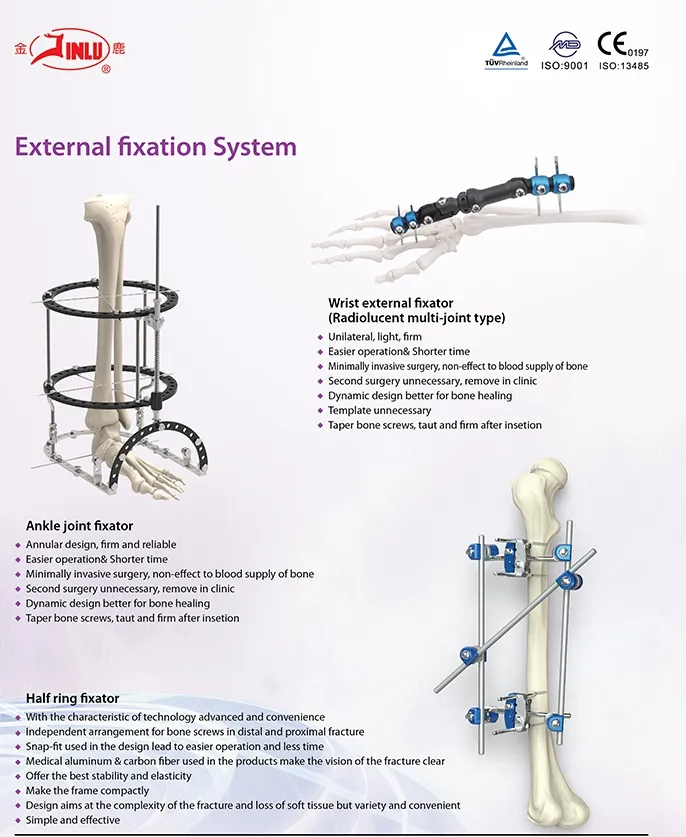
Our catalog includes Metal Bone Plates and Screw Systems, Metallic Cannulated Bore Screws, Metallic Interlocking Intramedullary Nails, Spinal Fixation Devices, and External Fixation Devices.
With over 60 years of experience in R&D and manufacturing, we offer a proven track record of reliability, a strict quality control system with CE/ISO/GMP certifications, and professional technical support.
We accept various delivery terms including FOB, CIF, EXW, and Express Delivery. Payments can be made via T/T, L/C, Western Union, or Escrow in USD, EUR, or CNY.
Our products are distributed globally, with a strong presence in Southeast Asia, the Middle East, South America, Africa, Central America, and parts of Europe.