














| Material | Titanium, Steel, Plastic | Feature | Disposable |
|---|---|---|---|
| Certification | CE, FDA, ISO13485 | Group | Adult and Children |
| Classification | Class II | Power Source | Manual |
| Package | Carton Packaging | Specification | 20X15X6CM |
| HS Code | 9018909919 | Capacity | 50000 pieces/Year |
DESCRIPTION: Surface skin suture, widely used in general surgery, tumor surgery, cardiothoracic surgery, obstetrics and Gynecology, burn surgery, bone surgery, emergency and field rescue. Applicable to human trauma and incision suture of surface layer skin during the surgery operation.
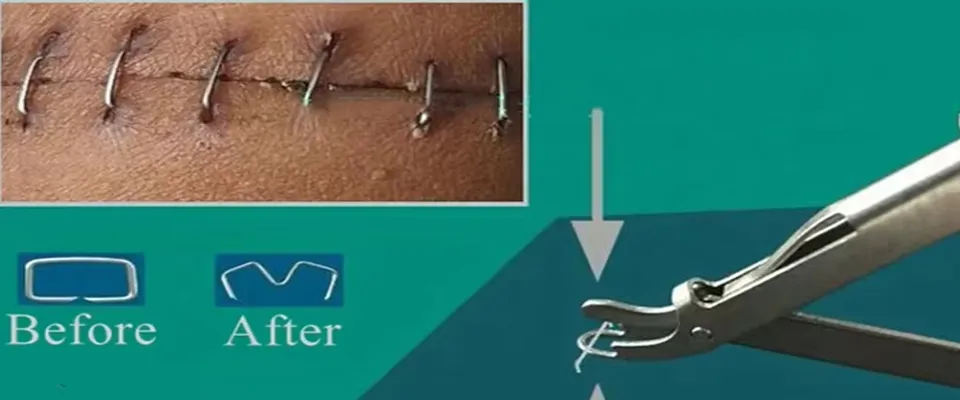
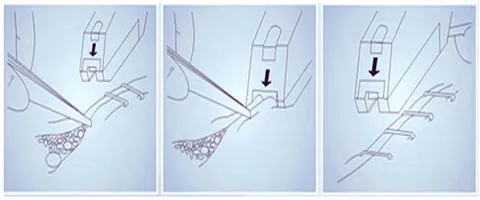
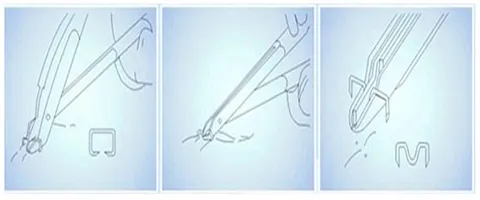
| Product Name | Disposable 35W 35R Skin Stapler Remover Surgical Operation 316L Skin Stapler | ||
| Model | 7.5X4.0 and 5.3X3.3 | Material | ABS, Stainless, Titanium |
| Validity | 3 Years | Certificate | CE, ISO |
| Packing | Packed in Color Box | Service | Online Technical Support |

Specifically configured split staple devices allow for splitting staples without pain. The simple and reasonable design makes it easy to operate in various surgical environments, significantly reducing the workload of healthcare providers.








Quality Assurance: During the warranty period, repair costs arising from product quality issues are fully covered.
Customer Support: Our team provides technical assistance via multiple platforms and conducts regular follow-ups to ensure satisfaction and offer maintenance guidance.

Specialized in R&D, manufacturing, and sales of endoscopic microport medical devices and various staplers. We integrate advanced technology with rigorous research to optimize product structure and maintain high quality standards. Our mission is to safeguard patient health through continuous innovation and a commitment to excellence in the medical device industry.