














| P/N | Product Name | Material | Size |
|---|---|---|---|
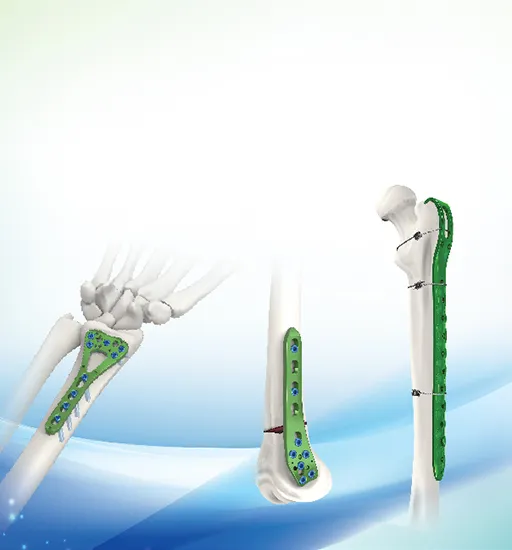
| 31102L/R | Multi-axial Distal Radius Palm Locking plate III (Left and Right types) | Pure Titanium | 2 holes L/R |
| 31103L/R | 3 holes L/R | ||
| 31104L/R | 4 holes L/R | ||
| 31105L/R | 5 holes L/R | ||
| 31106L/R | 6 holes L/R | ||
| 31107L/R | 7 holes L/R | ||
| 31108L/R | 8 holes L/R |






Established in 1958, we are specialized in the development, manufacture, and sales of orthopedic implants and instruments. As a key enterprise for national medical apparatus, the product line covers Spine pedical screws, Interlocking nails, Locking plates/screws, trauma plates/screws, cannulated screws, external fixators, hip prosthesis, and surgical instruments.
Our facilities include advanced imported CNC machinery, high-precision digital control systems, and a 100,000 Grade axenic purification chamber to ensure high-quality standards. We have achieved ISO 9001 quality system certification and our products have received numerous national and provincial science and technology awards.
We adhere to the development tenet of "creating high quality with science and technology". Our core focus remains on perfect design, innovative technology, and top-ranking products to serve the medical society.
