




1 / 5










Surgical Instrument Orthopedic Joint Power Drill Saw Hip Reamers
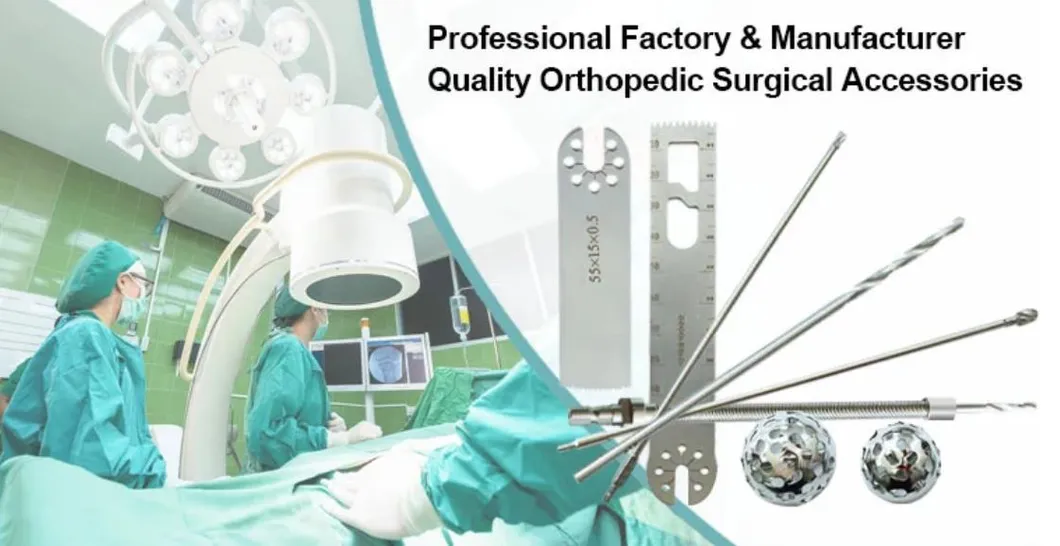
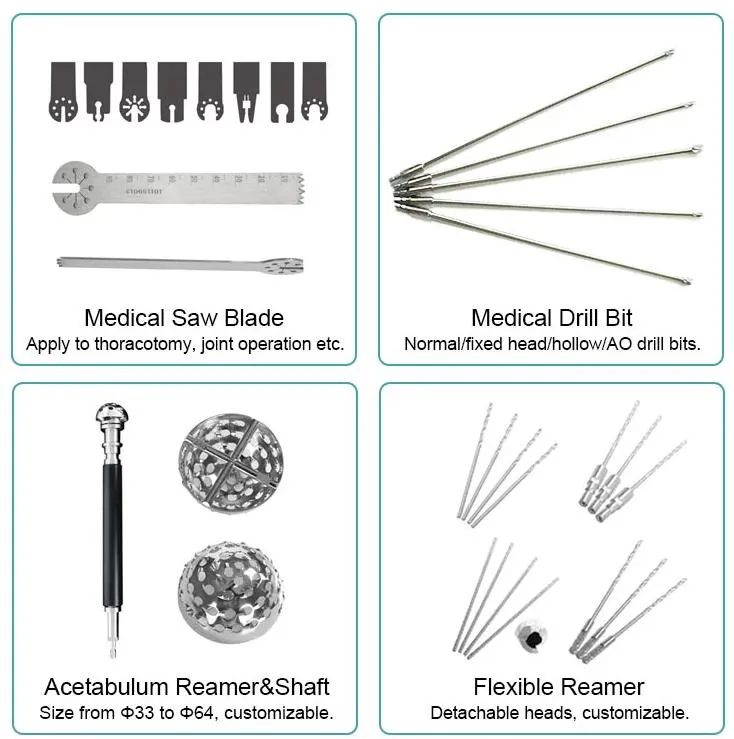





| 1. The device is forbidden to be demolished by non-professionals. |
| 2. Products are chargeable, compatible with 110V/60Hz - 220V/50Hz power sources. |
| 3. The battery must be removed from the handpiece when not in use to avoid automatic discharge. |
| 4. The battery cannot be disinfected. Ensure the battery compartment is dry after sterilization. |
| 5. Clean saw blades and drill bits with a purifier and keep dry post-operation. |
| 6. Maximum sterilization temperature is 135°C; exceeding this may damage the motor. |

