



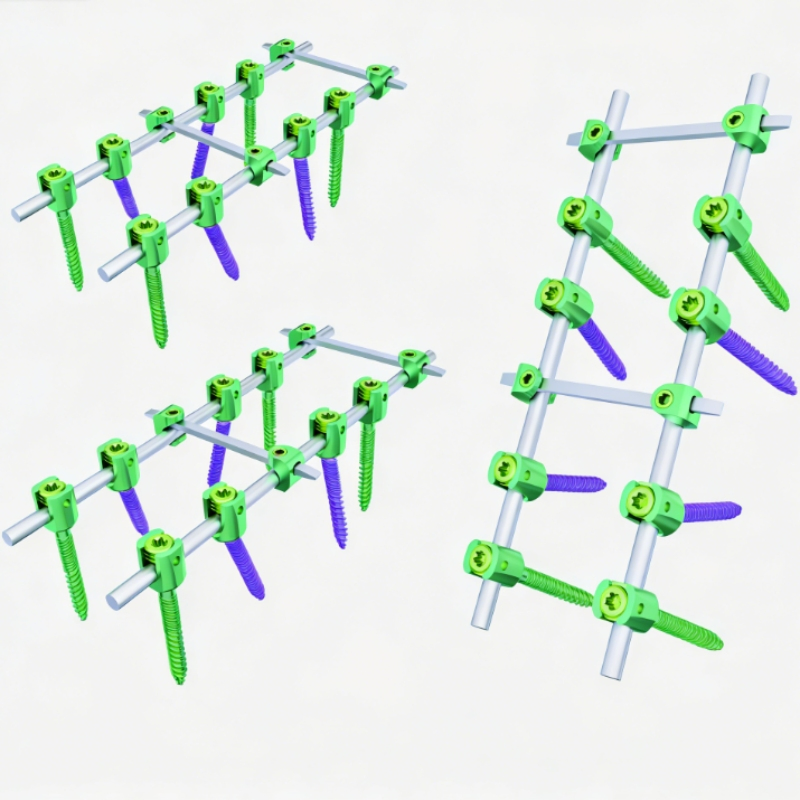













The Spine Pedicle Screw System is a comprehensive spinal fixation platform designed to deliver secure, segmental stabilization across the thoracic, lumbar, and sacral spine. Engineered for strength, adaptability, and ease of use, this system supports a wide range of spinal procedures, from degenerative corrections to complex deformities and trauma reconstruction.
Featuring polyaxial screw heads and a streamlined locking mechanism, the system simplifies rod placement and reduces intraoperative challenges. Constructed from medical-grade titanium alloy, it provides excellent biomechanical performance, biocompatibility, and compatibility with advanced imaging technologies.
| Product Name | Spine Pedicle Screw System |
| Material | Medical-Grade Titanium Alloy (Ti-6Al-4V ELI) |
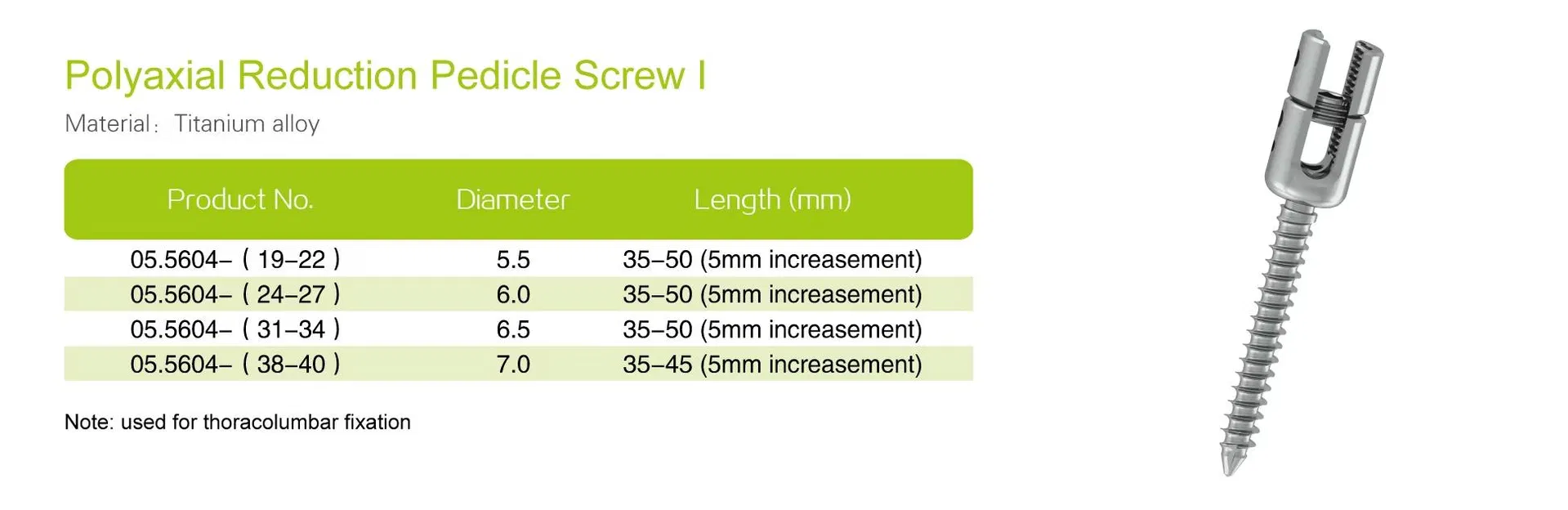
| Screw Diameters | 5.5 mm, 6.0 mm, 6.5 mm, 7.0 mm |
| Screw Length Ranges | 30–45 mm, 35–50 mm, 35–45 mm |
| Screw Types | Polyaxial, monoaxial, reduction, cannulated options |
| Rod Diameter Compatibility | 5.5 mm, 6.0 mm, 6.35 mm |
| Locking Mechanism | Set screw, dual-thread locking, or top-lock cap |
| Instrumentation | Screwdrivers, awls, taps, guides, rod holders, reducers |
| Sterilization | Pre-sterilized (Gamma / EO), single-use |
| Regulatory | CE Marked, FDA Cleared (as applicable) |









Our company is specialized in developing, production and sales of orthopedic implants and instruments. The factory covers a ground space of 32,068 square meters. We have a strong group of engineers and technicians, satisfying customers' requirements with consistent quality and favorable service, based on advanced production technology and testing equipment imported from Germany.
The main products include: Spinal fixation systems, interlocking intramedullary nails, metal bone plates, metal bone screws and related operation instruments. We have passed ISO 9001 and ISO 13485 system certifications, and products have passed CE certification. We serve customers from over 50 countries across the Middle East, Africa, Southeast Asia, East Europe, Australia, and the Americas.



