📋
Basic Information
Model NO.:MIS Spine
Condition:New
Usage:Fracture Fixation
Using Site:Cervical
Type:Splint Fixation
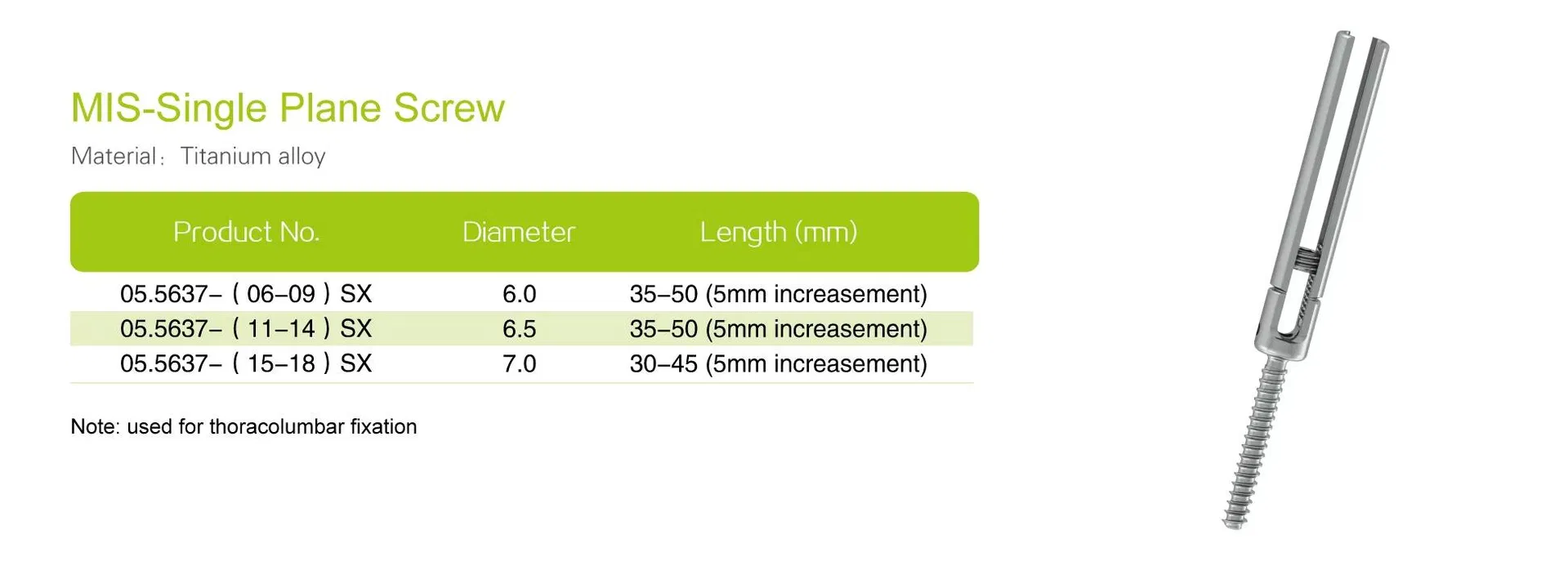
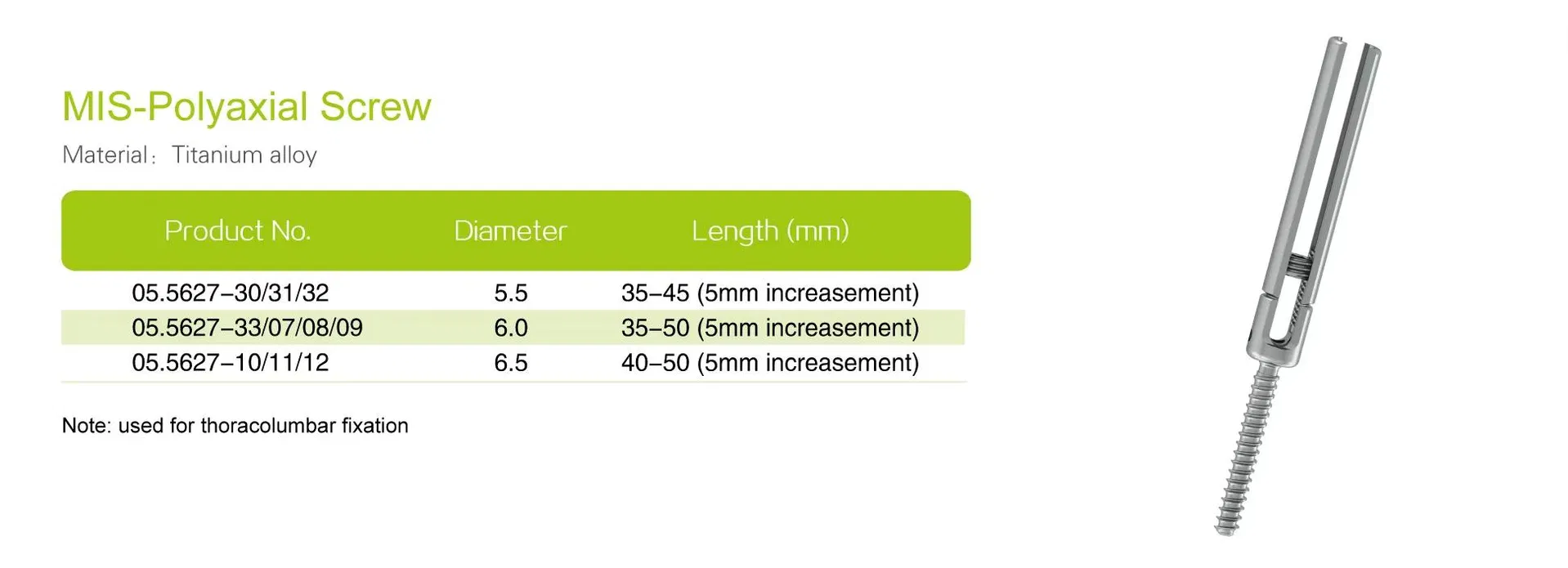
Material:Titanium Alloy
Screw Type:Polyaxial / Monoaxial
Size:3.5 / 4.0 / 5.5 / 6.0 / 6.5
Origin:China
Production:50,000 Pieces / Year
🔍
Product Description

The MIS Spine System is a specialized spinal fixation solution designed for minimally invasive surgical approaches. Engineered with precision and adaptability, the system provides robust stabilization while minimizing tissue disruption, reducing postoperative pain, and accelerating recovery.
Constructed from high-strength medical-grade titanium alloy, the implants offer excellent biocompatibility and durability, supporting long-term spinal fusion outcomes.
| Product Name | MIS Spine Fixation System |
| Material | Medical-Grade Titanium Alloy (Ti-6Al-4V ELI) |
| Screw Diameter | 5.5mm, 6.0mm, 6.5mm |
| Screw Length | 35–50mm (Varied configurations) |
| Screw Type | Cannulated, self-tapping, multiaxial or fixed-angle |
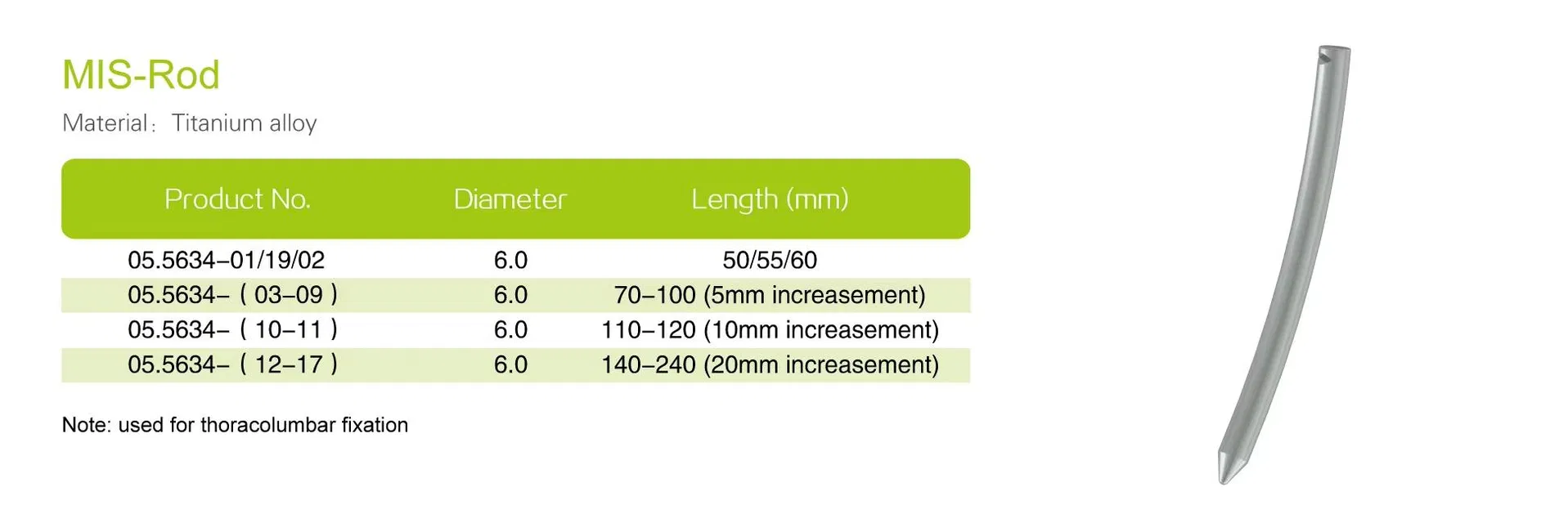
| Rod Diameter | Typically 5.5mm or 6.0mm |
| Sterilization | Pre-sterilized (Gamma or EO), single-use |
| Regulatory | CE Marked, FDA Cleared (as applicable) |
🏢
Manufacturing Excellence

Our manufacturing facility specializes in developing, producing, and selling orthopedic implants and instruments. The factory covers a ground space of 32,068 square meters. With a strong group of engineers and technicians, the company satisfies global requirements through advanced production and processing technology, utilizing high-end testing equipment.

We provide a comprehensive range of products including spinal fixation systems, interlocking intramedullary nails, metal bone plates, and surgical instruments. Our quality management system is certified under ISO 9001 and ISO 13485, with products carrying the CE certification.
📦
Packaging & Shipping

Products are packaged in sterile, procedure-specific trays with organized components to ensure safety and efficiency in the operating room. We export to over 50 countries across the Middle East, Africa, Southeast Asia, Europe, and the Americas.
❓
Frequently Asked Questions
How can I obtain a product sample?
Please provide the sample cost and express fee initially. We will refund the sample cost to you upon the placement of your first official order.
What is the typical sample lead time?
For existing inventory items, samples can be dispatched within 3 days.
Do you offer OEM or custom branding services?
Yes, we can print your logo on both the products and the packaging, provided the Minimum Order Quantity (MOQ) is met.
Can the color of the orthopedic implants be customized?
Yes, the color of the products can be customized according to your requirements if the MOQ is satisfied.
How do you ensure product quality control?
We implement strict detection protocols during production and conduct thorough sampling inspections before shipment to ensure intact packaging and high-quality standards.
What materials are used in the MIS Spine System?
The system is manufactured from medical-grade Titanium Alloy (Ti-6Al-4V ELI), known for its high strength and excellent biocompatibility.