














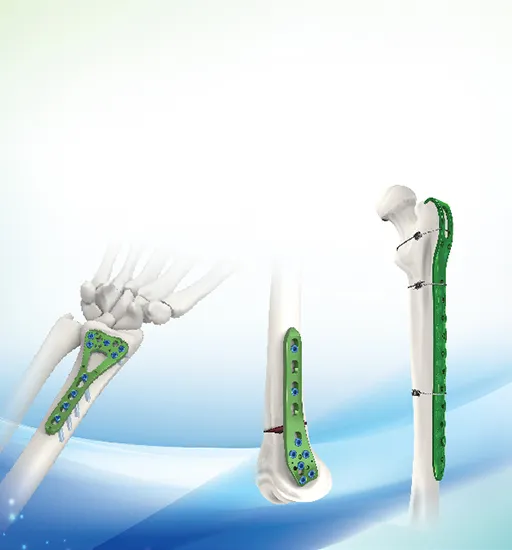
| Proximal Femur Neck with Hook Locking Plate 238 | ||
| P/N | Material | Holes |
| 23804-23812 | Titanium | 4, 6, 8, 10, 12 holes |
| Use 5.0 locking screw, 5.0 cortical self tapping screw | ||







Established in 1958, we are engaged in the development, manufacture and sales of orthopedic implants and instruments. We are also the key enterprises for national medical apparatus and a director company of the industry association. Our main product lines cover Spine pedicle screws, Interlocking nails, Locking plates/screws, trauma plates/screws, cannulated screws, external fixators, hip prosthesis and surgical instruments.
Our products are characterized by multi-varieties and complete specifications. All medical apparatus have passed the approval and authentication of national and provincial superintendent offices. Advanced equipment, including imported CNC, high precision digital control machines and milling machines, as well as a 100,000 Grade axenic purification chamber, contribute to the high quality of our products.
We adhere to the development tenet of "create high quality with science and technology, serve the society with love". We pursue perfect design, innovative technology, top-ranking products and complete after-sales service.
